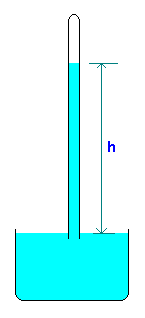
A barometer is a device for measuring atmospheric pressure. A simple barometer consists of a tube more than 30 inch (760 mm) long inserted in an open container of mercury with a closed and evacuated end at the top and open tube end at the bottom and with mercury extending from the container up into the tube. Strictly, the space above the liquid cannot be a true vaccum. It contains mercury vapor at its saturated vapor pressure, but this is extremely small at room temperatures (e.g. 0.173 Pa at 20oC).
The atmospheric pressure is calculated from the relation Patm = ρgh where ρ is the density of fluid in the barometer.
Last Modified on: 14-Sep-2014
Chemical Engineering Learning Resources - msubbu
e-mail: msubbu.in[AT]gmail.com
Web: http://www.msubbu.in